



Start Refreshed
End the Day Calm
The best nature has for Calm & Sleep.
Find out why people are raving about Natural Spectrum CBDA Oil

Over 10,000 Servings Sold!
Unlock Your Best Self!

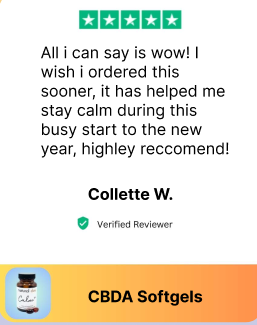
“They’ve really been helping me with stress and anxiety. What i like about them so much is you dont just have to take them at night, you can take them during the day.”
Brittany F.
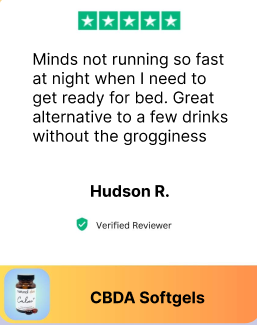
“I take 1 at night and it helps me get a good nights rest.”
Chelsea C.

Chloe B.
-get started
CBDA Gummies with Delta 8 - Vegan Tropical 900mg
$59.99 — orCBDA Gummies - Vegan Tropical 900mg
$32.99 - $59.99 — or $32.99 - $44.99 / monthRelaxing CBDA Breath Mints with Delta 8 THC - 900mg
$29.99 — orRefreshing CBDA Breath Mints - 500mg
$24.99 — orCBDA Tincture - Orange Zest 500mg
$44.99 — orCBDA Tincture - Orange Zest 1000mg
$59.99 — orCBDA Tincture - Mint 1000mg
$59.99 — orCBDA Tincture - Mint 500mg
$44.99 — orCBDA Oil for Peak Relaxation
Over 500% more potent than CBD without the high!

Promote Calmness & Relaxation*
Experience serenity with each gummy. Our Natural Spectrum CBDA formula effortlessly melts away life's tensions, guiding you to a state of relaxation.

Reduce Daily Stress*
Stay cool and composed, even on your toughest days. Our CBDA-powered blend helps you navigate life's twists and turns, putting stress in the rearview.

Improve Healthy Sleep*
Reclaim your nights. Our gummies, packed with the natural benefits of Natural Hemp Cannabinoids, pave the way for deep, rejuvenating sleep.

Lessen Fatigue & Recharge*
Recharge and rejuvenate! With Natural Spectrum CBDA at its core, our gummies kick fatigue to the curb, re-energizing you for whatever the day throws your way.
Why Everyone is Switching to CBDA Oil
CBDA is the most potent calming cannabinoid on the market
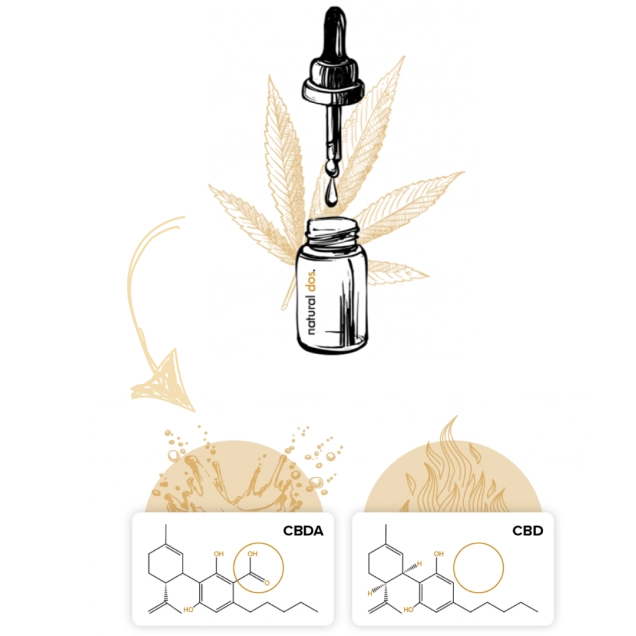
CBDA (cannabidiolic acid) is naturally made by hemp, whereas CBD is a byproduct of conventional manufacturing

A Natural Spectrum CBDA Oil is captured, and has over 500% better absorption vs CBD

CBDA Oil offers the natural calming and refreshing power of real hemp
Our Promise
100 Day Money Back Guarantee
We’d be shocked if you didn’t feel the effects, and enjoy the benefits of the Natural Spectrum CBDA Gummies. But in the highly unlikely event that you’re not happy in any way — we’ll give you every cent back. No hassle. No hoops to jump through.
You’re protected by our 100-Day 100% Moneyback Guarantee. Email us at sales@naturaldos.com and we’ll return every dollar you paid.

Why Have I Not Seen More CBDA Products?

Regular hemp extraction destroys CBDA, making CBD.

No CBD company wants to admit CBDA is better since they sell a lot of CBD.

Only Natural Dos scientists developed new technology to make a Full Spectrum Oil
Why Natural Spectrum CBDA Oil?
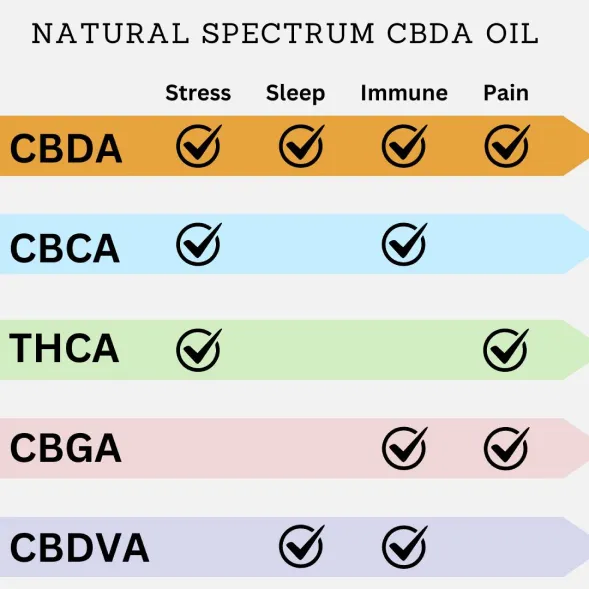
Natural Spectrum CBDA Oil contains all the Acidic Cannabinoids
Natural Dos is the only company that purifies ALL the naturally occurring acidic cannabinoids
When all the cannabinoids are present absorption and health benefits are enhanced even further!
BREAKING NEWS
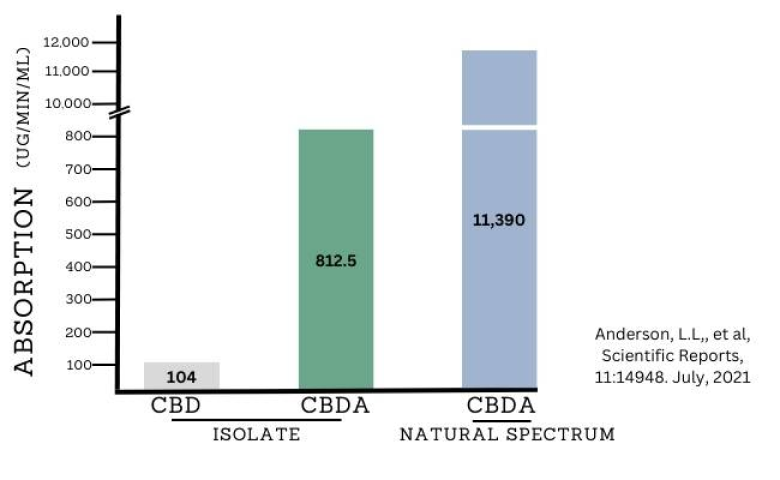
Highlights
CBDA combined with other cannabinoids shows over 1000% better absorption vs CBD
CBDA has the most bioavialability of any cannabinoid

Made in USA
Our hemp is grown in Colorado and purified by our scientists in our laboratory in Birmingham, AL. Unlike most supplement and hemp companies we make our own active ingredients to ensure highest quality is maintained.

Third Party Lab Tested
Our rigorous testing requirements on all products elevates us to the top, and is even more stringent than most stuff you buy in the grocery store. See our CoA page for all results.

Natural Ingredients
We source the best ingredients to go along with our Natural Spectrum CBDA, such as organic MCT oils, natural fruit extracts, and organic cane sugar.

Gluten Free
Many people may not know they are allergic to gluten, which can come as mild irritation and inflammation in the gut when consumed. None of our products contain gluten or are exposed to it during manufacturing.
CBDA (cannabidiolic acid) is the parent compound of CBD, and the most abundant cannabinoid made by hemp. Almost all cannabinoids are naturally made in the “acidic form”, meaning they contain a carboxyl group, and are typically converted to their neutral forms, think CBD, during conventional purification processing. CBDA, like other acidic cannabinoids, have superior absorption and only recently are being investigated for their potential therapeutic properties.
CBDA (cannabidiolic acid) has between 500% to over 1000% better absorption than CBD (cannabidiol). Absorption is critical to the strength of any compound, and determines its bioavailability which refers to how much gets into the bloodstream. The natural absorption for CBDA and other acidic cannabinoids are higher than their neutral forms (e.g. CBD), and can be enhanced when combined in a full spectrum product.
CBDA has been tested for a range of benefits, such as anti-viral, stress and anxiety, nausea, sleep, and pain and inflammation. A large number of its benefits come from its unique combination of targets in the inflammatory process, and serotonin and endocannabinoid system.
Users typically report more relaxation and calm, with some finding relief for different aches and pains. Some users have found that it can help with sleep if they have an overactive brain that doest stop.
The hemp plant (and all cannabis sativa species) make cannabinoids in the “acidic form”, which means cannabinoids with carboxyl groups. Thus, CBDA (cannabidiolic acid), is the most abundant cannabinoid in the hemp flower, and is the parent compound to CBD (cannabidiol). During conventional purification processes, the heat involved converts CBDA to CBD in a process termed decarboxylation (the CBDA loses the carboxyl group).
CBDA dosage is a common question because of its enhanced absorption. A significant issue in the hemp/CBD space is the low absorption, and we think the typically recommended dose range is too low to get substantial benefits. This problem is addressed by using CBDA which has over 5X better absorption, and over 10X better absorption in full spectrum products like Natural Spectrum CBDA from Natural Dos.
Yes, we have a 100 day 100% money back guarantee if you are not satisfied with your Natural Dos CBDA product. Just let us know within the first 100 days of purchase.
Have We Met?
Natural Dos is the first company to create a Natural Spectrum CBDA Oil with its proprietary purification technology.
Join us for the next evolution of natural hemp relief.















